Biologics Modular, LLC has received its DD2345 certification documents.
...More »Here are a couple of short videos showing how changes made to a simple duct into a unidirectional high efficiency particulate air (HEPA) filter can improve flow efficiency.
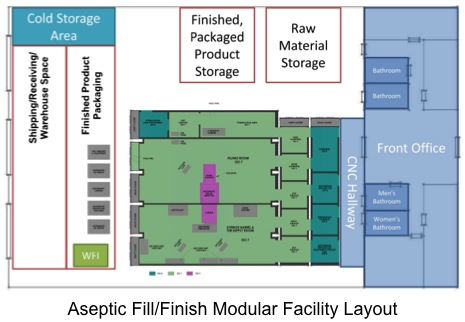
...More »Constructing a cGMP-compliant facility overseas can be difficult, so at Biologics Modular we solve that problem for you. Your modular, cleanroom facility is constructed under our closed-manufacturing platform, which means consistency and convenience in quality and design. Our platform includes the following validations and amenities: IQOQ, SAT, FAT, HVAC, electrical, security, monitoring, commissioning, qualification, and third party certification. Following construction, we will transport and assemble your pre-validated, modular facility anywhere in the world.